WHO Model Formulary: Global Standards for Essential Generics
 Apr, 18 2026
Apr, 18 2026
Imagine a world where a life-saving antibiotic or a blood pressure medication is available, but only if you can afford a price tag that exceeds a family's monthly income. This gap is exactly what the WHO Model List of Essential Medicines is designed to close. It isn't just a list of drugs; it's a global blueprint that ensures the most critical medications-specifically generics-are safe, effective, and affordable for everyone, regardless of where they live.
What Exactly is the WHO Model List?
At its core, the WHO Model List of Essential Medicines (often called the EML) is a technical guide developed by the World Health Organization. It identifies medicines that satisfy the priority healthcare needs of populations globally. First launched in 1977, the list is updated every two years to reflect new medical evidence and emerging health threats.
It is important to understand that the EML isn't a "formulary" in the way a US insurance company or a specific hospital uses the term. While a commercial formulary might focus on cost-sharing tiers or insurance coverage, the WHO list focuses on public health relevance. It tells countries: "These are the medicines your health system must have in stock at all times to keep your people alive and healthy."
The list is split into two main parts. The core list contains the absolute essentials-the most safe and cost-effective drugs for the most common conditions. The complementary list includes medicines that might require specialized equipment or specialist training to administer, such as certain advanced cancer treatments.
The Gold Standard for Essential Generics
Why is there such a heavy emphasis on generics? Because they are the key to affordability. In the 2023 update, about 46% of the listed medicines are generics. By promoting these versions instead of expensive brand-name drugs, the WHO helps countries slash their pharmaceutical spending.
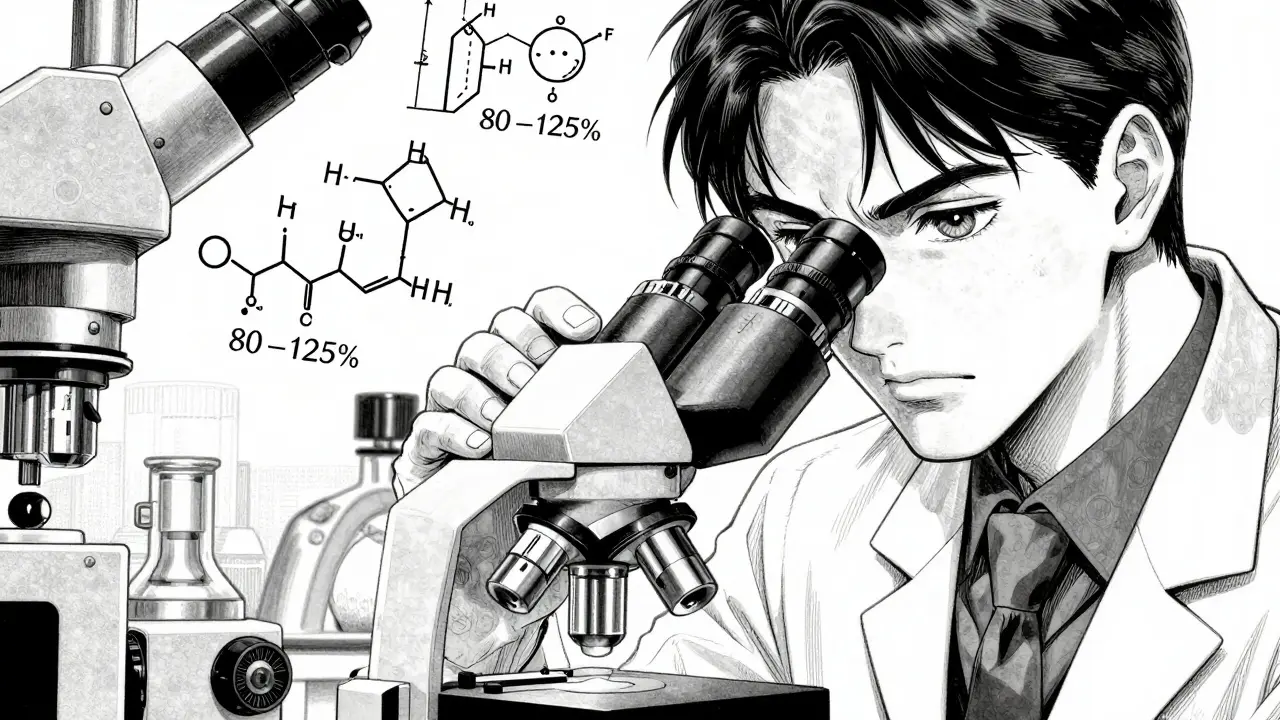
But not all generics are created equal. To prevent substandard or fake drugs from entering the supply chain, the WHO uses a rigorous process called WHO Prequalification. This is a quality-assurance service that ensures a generic drug is bioequivalent to the original brand-name version. Specifically, they look for 80-125% confidence intervals for AUC and Cmax ratios. For high-risk drugs with a narrow therapeutic index, these limits are even tighter (90-111%) to ensure patient safety.
| Feature | WHO Model List (EML) | Commercial/Insurance Formulary |
|---|---|---|
| Primary Goal | Universal Public Health Access | Cost Management & Coverage |
| Selection Criteria | Evidence of efficacy, safety, & cost | Contract negotiations & rebates |
| Structure | Core vs. Complementary | Tiered Cost-Sharing (Tiers 1-5) |
| Focus | Essential Generics | Brand vs. Generic mix |
How Medicines Actually Get on the List
Getting a drug onto the EML isn't a matter of lobbying; it's a matter of data. The WHO Expert Committee on Selection and Use of Essential Medicines reviews hundreds of applications using a strict scoring system. If a drug doesn't hit a 7/10 in every single category, it doesn't make the cut.
- Public Health Relevance (30%): Does it treat a disease with a high burden? (Usually requires at least 100 cases per 100,000 people).
- Efficacy and Safety (30%): Is there high-level evidence from randomized controlled trials?
- Comparative Cost-Effectiveness (25%): Is the cost per quality-adjusted life year (QALY) reasonable relative to the local GDP?
- Programmatic Feasibility (15%): Can it actually be stored and administered in a real-world clinic?
This evidence-based approach means that 94% of inclusions are backed by systematic reviews. It removes the guesswork and prevents wasteful spending on "trendy" drugs that don't actually provide a significant clinical advantage over cheaper generics.
Real-World Impact: Does It Actually Work?
The numbers suggest a resounding yes. Countries that align their national lists with the WHO standards often see pharmaceutical expenditure drop by 23% to 37%. For example, in Ghana, adopting these principles helped reduce out-of-pocket medicine costs by 29% between 2018 and 2022.
The most dramatic success story is in HIV treatment. By pushing for generic antiretrovirals, the median price dropped from over $1,000 per patient per year in 2008 to just $119 in recent years. This shift allowed the number of people receiving treatment to jump from 800,000 in 2003 to nearly 30 million by 2022. That is the power of prioritizing generics over patents.
However, the system isn't perfect. While the list is great, the supply chain can be a nightmare. In Nigeria, some surveys showed that while the national list was based on WHO standards, nearly 60% of essential medicines faced stockouts. The list tells you what you need, but it doesn't magically put the pills on the shelf.

The Future of Global Generic Standards
The WHO is currently evolving the list to keep up with modern medicine. We are seeing a move toward Biosimilars-which are essentially generic versions of complex biological drugs. The 2023 update already includes several monoclonal antibodies with specific bioequivalence ranges.
There is also a major push for pediatric health. In the past, many essential drugs only came in adult doses, forcing doctors to "guess" or manually split pills for children. Now, over 42% of the medicines on the list have age-appropriate formulations, up from 29% just a few years ago.
Looking ahead, the WHO is integrating the list with digital tools, like the Essential Medicines App, to help clinicians in remote areas make better prescribing choices. They are also focusing on "antibiotic stewardship," creating tiers to prevent the overuse of powerful antibiotics and stop the rise of superbugs.
Is the WHO Model List mandatory for all countries?
No, it is not mandatory. It serves as a technical reference. However, over 150 countries use it as the foundation for their own National Essential Medicines Lists (NEMLs) to ensure they are providing the most effective and affordable care.
What is the difference between a generic and a biosimilar on the WHO list?
A generic is a chemical copy of a small-molecule drug. A biosimilar is a version of a large, complex biological protein. Because biologicals are harder to copy exactly, biosimilars have different, slightly wider bioequivalence ranges (85-115%) compared to traditional generics.
How does WHO Prequalification help patients?
It acts as a seal of approval. When a generic is prequalified, it means the WHO has verified that the manufacturer meets strict quality standards. This protects patients from substandard or falsified medicines, which is a major risk in low-income regions.
Why are some newer drugs not included in the WHO list?
The WHO prioritizes cost-effectiveness and public health burden. If a new drug is only slightly better than an existing generic but costs 100 times more, it likely won't be added. The goal is to maximize the health of the most people with the limited resources available.
Does the WHO list affect medicine prices in the US?
Directly, very little. US domestic formularies are managed by private insurers and government programs like Medicare. However, the WHO list heavily influences global procurement for UN agencies and international aid, which can drive down global prices for generics.
Next Steps for Health Systems
If you're a healthcare administrator or a policymaker, simply adopting the list isn't enough. To make it work, you need to focus on three things: first, ensure your procurement policies prioritize WHO Prequalified generics to avoid counterfeit drugs. Second, invest in supply chain infrastructure to reduce the "stockout days" that plague many hospitals. Finally, train staff on generic substitution-teaching them that a prequalified generic is therapeutically equivalent to the brand-name version can save a health system millions of dollars.
